














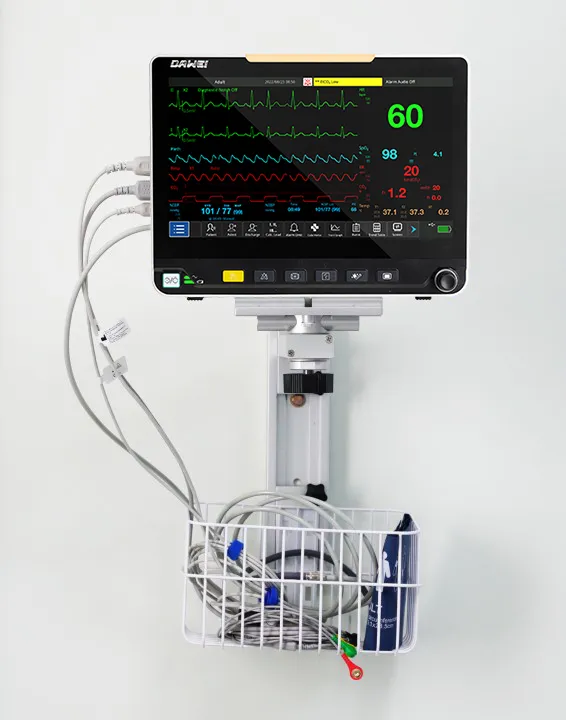
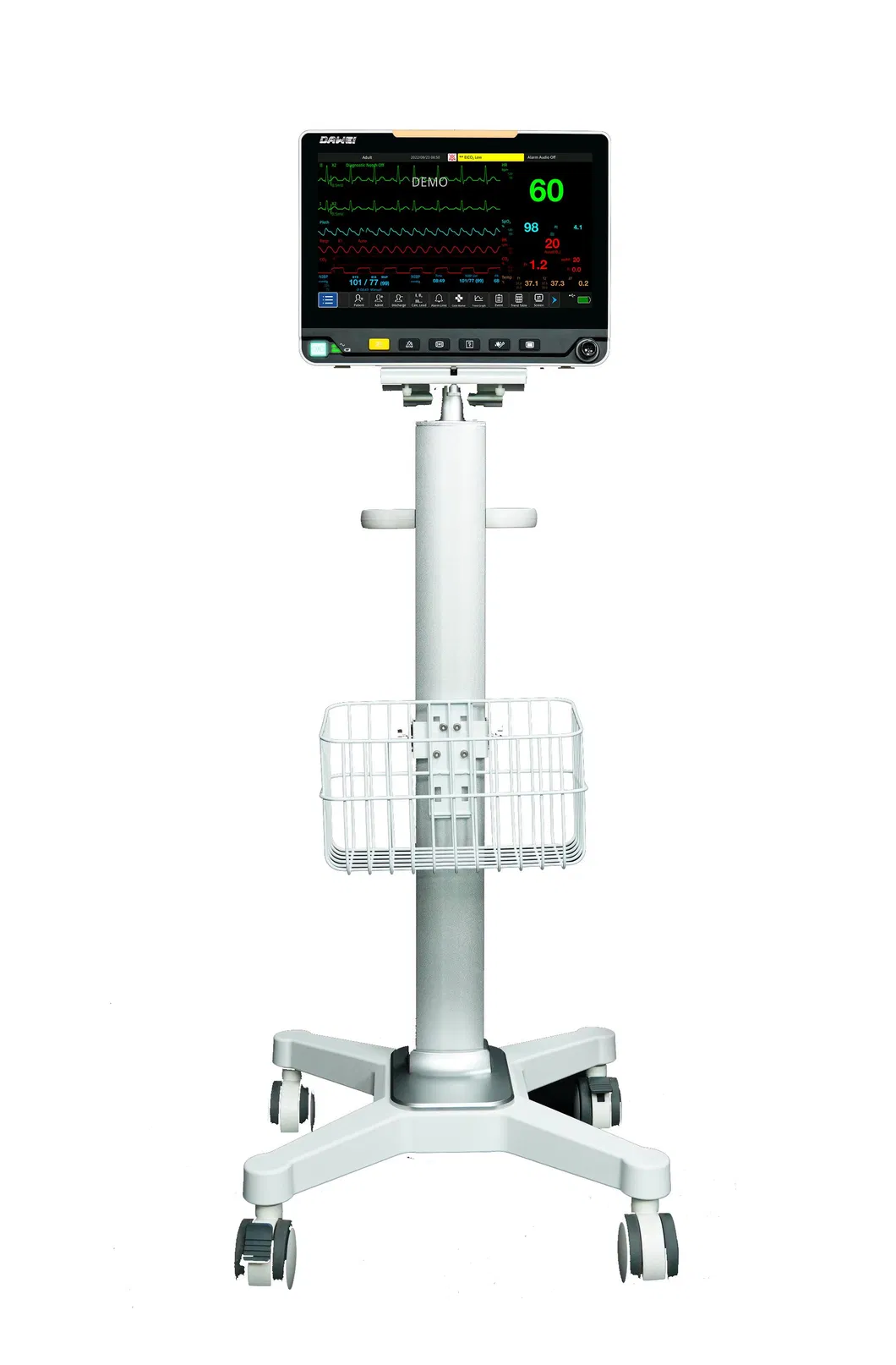
Patient Monitoring System ICU Multi-Parameter Patient Monitor
| Standard Configuration | Optional Configuration |
|---|---|
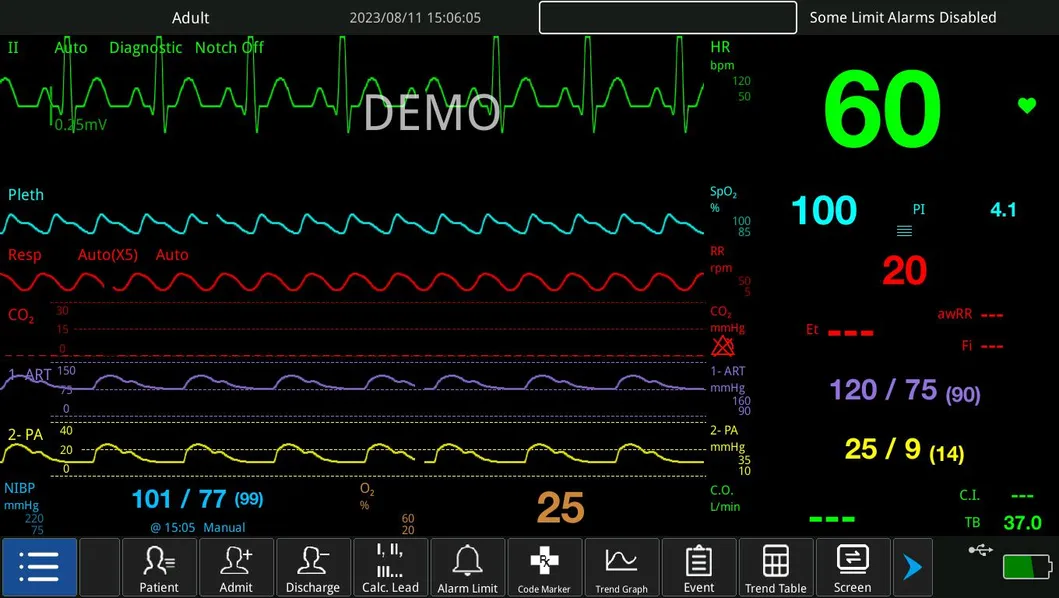
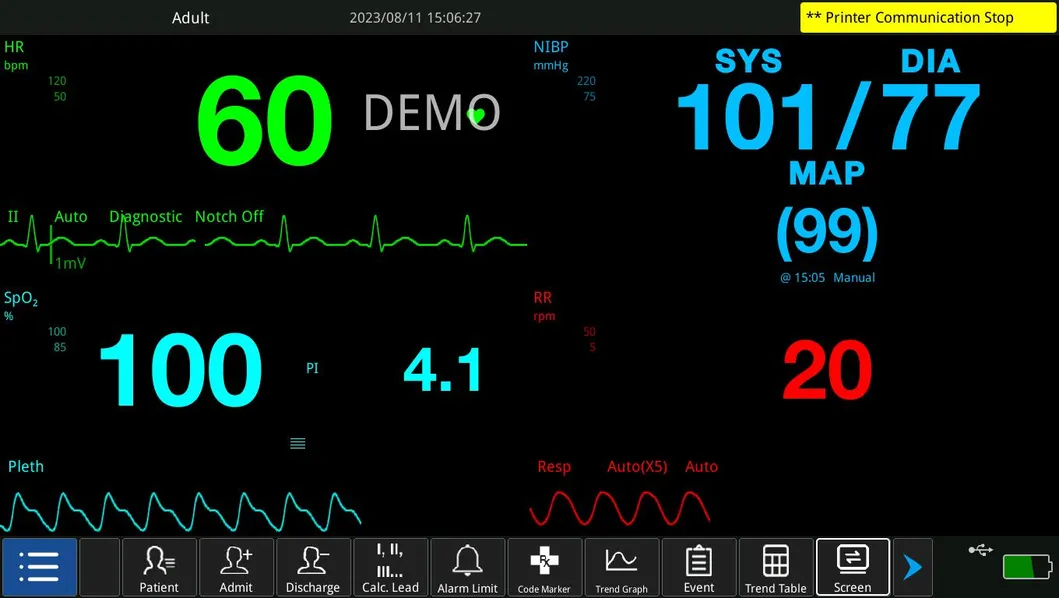
| ECG, RESP, NIBP, SpO2, PR, TEMP | IBP, CO, ICG, EtCO2, AG, RM, BIS |


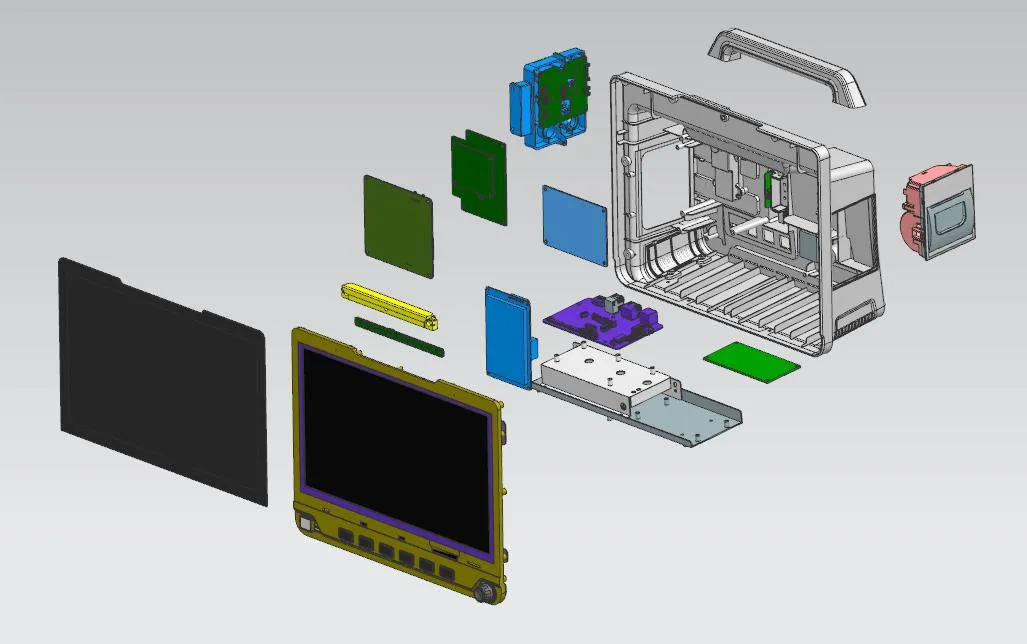
OEM/CKD Service Available



ISO 13485 certification is an international standard for the medical device industry worldwide. This certification emphasizes the establishment and maintenance of a quality management system, aiming to ensure that products meet rigorous industry requirements.
Our production processes undergo rigorous monitoring to ensure product consistency and compliance with regulatory requirements. We actively adopt innovative technologies and materials to continuously optimize product performance and reliability.
Our manufacturer is a well-established domestic medical device provider recognized as a national high-tech enterprise. Since 2006, the company has been dedicated to the innovative development and manufacturing of ultrasound imaging, digital electrocardiographs, and digital radiology products.
The product portfolio consists of over 50 models in five major categories, including color Doppler ultrasound systems, black and white ultrasound machines, ECG machines, patient monitors, and digital radiography equipment. All products have obtained ISO 13485:2012 and European CE certification.



Our team consists of experienced technical experts capable of responding promptly and resolving issues efficiently. We offer:
